The following diagram shows the GAMP guides in relation to the automation landscape as define in ISA-95.
see the Interactive view
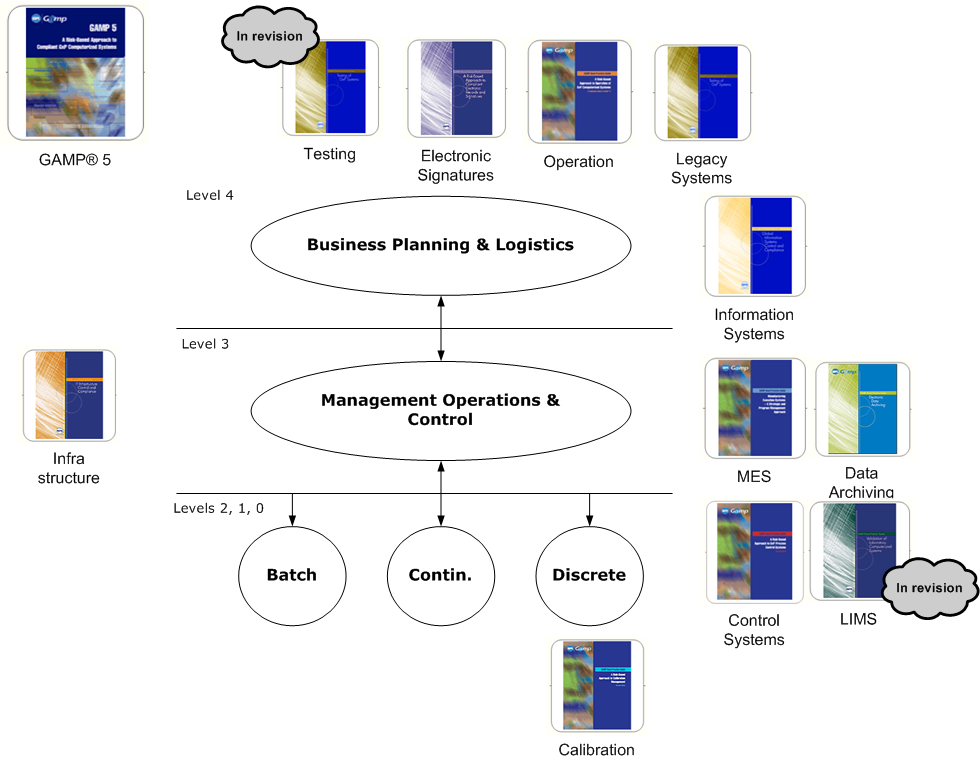
The following diagram shows the GAMP guides in relation to the automation landscape as define in ISA-95.
see the Interactive view

"Establishing documented evidence which provides a high degree of assurance that a specific process will consistently produce a product meeting its pre-determined specifications and quality attributes"
At this web site we concentrate on Validation of Automation systems. The best way to start Validation for an automation system is to buy the GAMP ® guide and take a training in using GAMP ®.
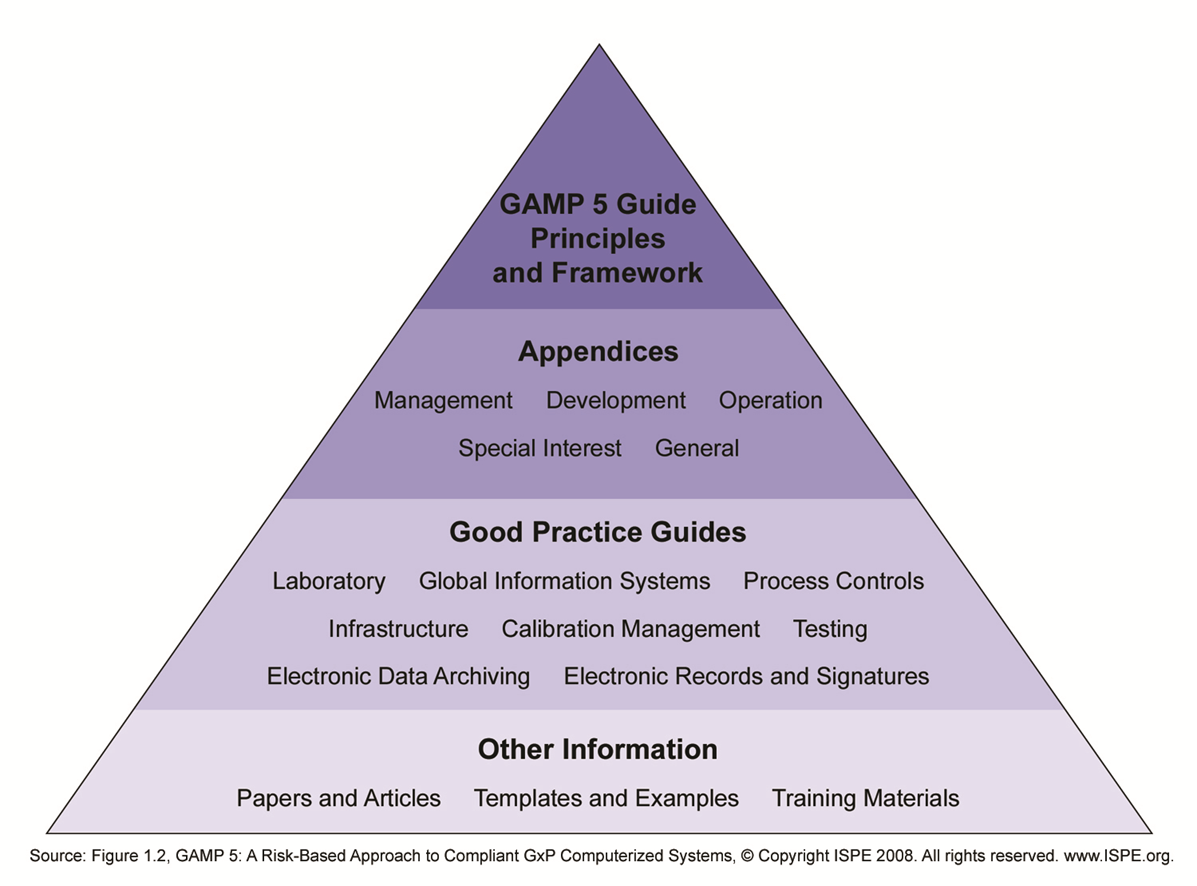
The GAMP 5 ® guideline is published by the ISPE (International Society for Pharmaceutical Engineering) in 2008 and provides guidelines to develop and maintain an automation system in the regulated industries. GAMP ® promotes a life cycle approach for an automation system. The life cycle is divided into the Project phase, Operation phase and Retirement phase.
For specific type of systems or validation subjects ISPE published Good Practice Guides:
The Good Practice Guides may be used in addition to the GAMP ® document.
Within the ISPE special GAMP communities are operational. For the Belgium, Netherlands and Luxemburg area you can participate in the GAMP Benelux Community of Practice.
created with
Joomla Page Builder.